 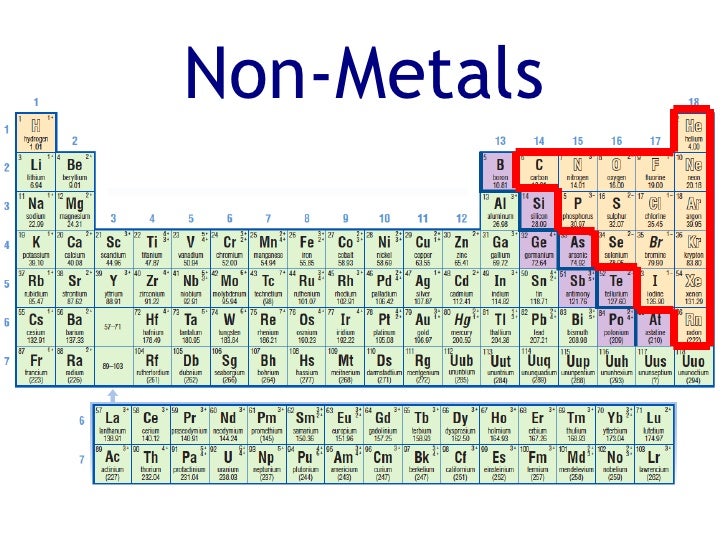
Now what do you think the properties of nonmetals are going to be? Well generally speaking, they're going to be the opposite of this. There's exceptions to this, they are solid at room temperature. Which is just a fancy way of saying that you canīend them without breaking. Ones that are in between, what are the properties of metals? Well, generally speaking, What are the nonmetals and maybe what are the The periodic table of elements what are the metals and Going to think about things as metals and as nonmetals. I'm going to introduce a broad classification of the elements, and in general, we're just To dig a little bit deeper and talk about the types of bonds that are likely to be formedīetween different elements. Idea of bonds between atoms, and we talked about the types of bonds, ionic, covalent and metallic. In a previous video, we introduced ourselves to the We can't do anything to change the polar covalent bond into an ionic bond because it's the identity of the elements which is determining the type of bond. So hydrogen and oxygen do not form an ionic bond simply because they do not have a large enough electronegativity difference. 
This would place the hydrogen oxygen bond in the polar covalent range instead of the ionic range. Hydrogen's electronegativity value is 2.20 while oxygen is 3.44, which result in a difference of 1.24 (3.44 - 2.20 = 1.24). Using the Pauling scale of electronegativity we need a difference greater than 2.0, x > 2.0. The very electronegative atom in an ionic bond will attract electrons to itself to such a degree that it removes electrons from the other, less electronegative atom. A large electronegativity difference means that a very electronegative atom was bonded with a not so electronegative atom. So it's unclear as of now what it is.įor an ionic bond to happen, the electronegativity difference between the two bonding atoms has to be large enough. Which would be a break from the trend of the group of unreactive gases at normal conditions. It's in the same group as the noble gases (which are nonmetals), but has been hinted that it may be solid under normal conditions and significantly reactive. So since so few atoms of Og have been created it's hard to say conclusively what it's properties are as of now. It was given the "-on" suffix since it belongs to group 18 along with the noble gases who all have suffixes of "-on". In 2016 is was officially decided to name it Oganesson (which the chemical symbol of Og) after Russian physicist Yuri Oganessian whose team helped synthesize superheavy elements 107 through 118. Ununoctium was a placeholder name for element 118 giving by the IUPAC in 1979 which was intended to be used until its discovery was confirmed since the people who discover an element gain the right to name it. Firstly, element 118 is no longer named ununoctium and has not been named so since 2016. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
